
So we can add/subtract in chemical equations, but its can't just simply make mathematical sense, it also has to make sense in a chemistry context. 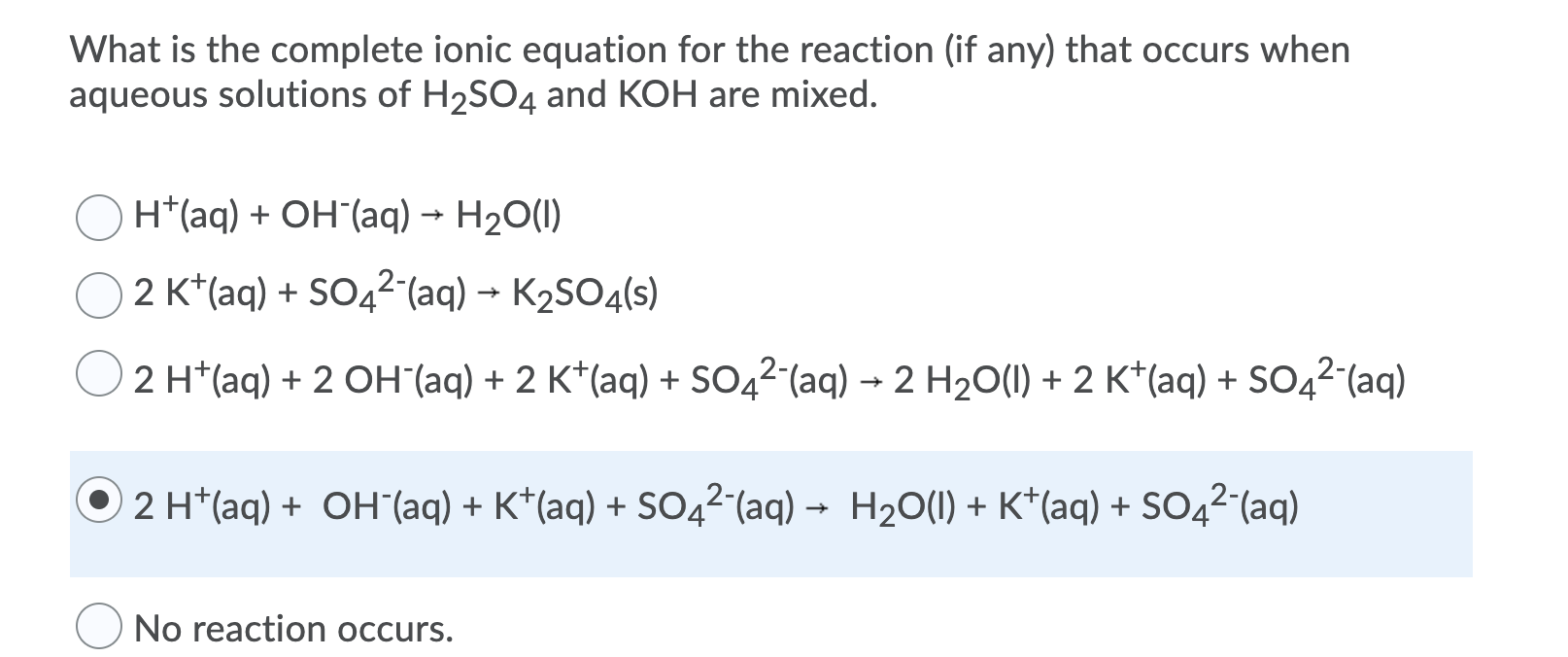
So it's not much that we're adding chemicals rather we're discovering the actual reaction occurring. However if it's an aqueous solution, these added chemicals are technically always present in the reaction solution and what we're actually doing is recognizing that some of them are actually part of the redox reaction. Now in this case where we're adding chemicals to the equation it may just seem like we're adding chemicals out of convenience to make the math work out. Lets first start with a complete chemical equation and see how the net ionic equation is derived. In redox reaction it's common to add water, H+, and OH- ion to the equations when balancing them. So since they're not participating in the reaction, subtract them is allowed because it doesn't affect the reaction if they're absent from the equation. Write down your observations molecular equation ionic equation and net. So these are ions which are present in the reaction solution, but don't really participate in the actual reaction (they don't change as a product compared to when they were a reactant). The chemical reaction: 8 Fe + S 8 FeS is a:Q1: What is an Ionic Equation. For ionic equations like these it's possible for us to eliminate, essentially subtract out, spectator ions from an equation. Mathematically it's completely acceptable to do so, however we have to consider the actual chemical makeup of our reaction if we do so. In sodium chloride, the ion-dipole bonds release more energy than was required to break the ionic bonds so it is soluble, but for silver chloride the ion-dipole bonds release less energy than is required to break the ionic bonds so it is insoluble. Break apart the aqueous compounds into ions but leave. If more energy is required to break the bonds than is released during bond formation, then the solid is insoluble. Lets write the net ionic equation for the reaction above: Pb(NO3)2(aq) + 2KI(aq) 2KNO3(aq) + PbI2(s). This is known as the complete ionic equation: Ag + (aq) + NO 3 (aq) + K + (aq) + Cl (aq) AgCl (s) + K + (aq) + NO 3 (aq) A final way to represent a precipitation reaction is known as the net ionic equation. For the dissolution process to be successful, we must release more energy than is required to break the bonds. This reaction can be also be written in terms of the individual dissociated ions in the combined solution. The same is true here.įor an ionic solid to dissolve, the solvent (in this case water) has to break the ionic bonds then surround the ions and form new ion-dipole bonds in a solvation shell. An energy input is required to break bonds, and energy is subsequently released when bonds are formed. The given equation shows the state of matter of the substances, otherwise you would of had to look at a solubility chart. The key is the energy associated with the dissolving process. Hi Jennifer Ionic equation show the substances that remain dissolved in solution and substances that will produce solid, liquid or gas. While both are sodium chloride and silver chloride are ionic compounds, sodium chloride is soluble and dissolves easily in water while silver chloride is insoluble and does not dissolve much into water. This reaction is represented by the molecular equation below. It’s the bottom line of the reaction.If we could zoom in on the contents of the reaction beaker, though, we wouldn't find actual molecules of AgNO 3 \text_4(aq) Na 2 SO 4 ( a q ) start text, N, a, end text, start subscript, 2, end subscript, start text, S, O, end text, start subscript, 4, end subscript, left parenthesis, a, q, right parenthesis. Notice how the net ionic equation gives you the essence of the reaction: a precipitate was formed with Co and OH reacted. Like spectators at a sporting event, they are not directly involved and are therefore not part of the main attraction. NOTE: spectator ions are ions that appear on both sides of the chemical equation that are eliminated before the net ionic equation is written. This video covers, how to predict products, how to balance a chemical equation, how to identify the solubility of a compound, how to write a complete ionic e. The net ionic equation is what is left at the end of the reaction, after the spectator ions have been eliminated.NOTE: gases, solids, and liquids DO NOT dissociate and remain in their molecular forms! The complete ionic equation is the entire chemical equation with all aqueous substances dissociated into their respective ions.The molecular equation is the full balance chemical equation. Equations: Complete Molecular, Complete Ionic and Net Ionic 1) Balanced molecular: FePO4(s) + 3KC2H3O2(aq) -> Fe(C2H3O2)3(s) + K3PO4(aq) 2) Complete ionic. 
Typically you will be asked to further dissect a chemical equation by writing not only the molecular equation, but additionally the complete ionic and net ionic equations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
